Baretich Engineering, in collaboration with Technology Management Solutions, conducted an assessment of operational risks associated with the nation-wide Clinical Technology Services program for the Kaiser Permanente system.
Areas that were assessed ranged from patient safety and regulatory compliance risks to financial and liability risks. Risks were assessed across the entire medical equipment life-cycle.
The assessment process included more than 40 telephone interviews with a total of over 200 individuals. It also included four site visits across the country. Findings were evaluated in terms of impact and likelihood, which allowed prioritization of risk levels.
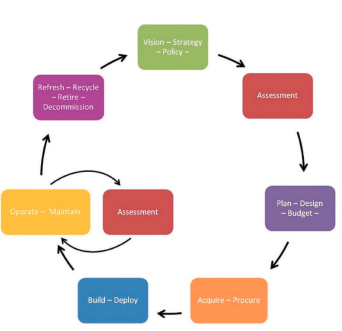
Extensive interaction with Kaiser Permanente leadership led to a detailed action plan that is in the process of implementation. A description of the project was published in the Spring 2015 edition of AAMI Horizons.
Baretich Engineering conducts numerous assessments of clinical engineering programs across the country, including most recently in Colorado, programs at Denver Health Medical Center and Children’s Hospital Colorado. We also conduct accreditation compliance assessments across the entire range of Joint Commission Environment of Care and Physical Environment standards, including most recently in Colorado, programs at Saint Anthony Medical Campus.
References
Carol E. Davis-Smith, Frank R. Painter, and Matthew F. Baretich (2015). Assessing Risk in the Kaiser Permanente Clinical Technology Program. Biomedical Instrumentation & Technology: Risk: How Do You Manage It Effectively?, Vol. 49, No. s1, pp. 60-64.
Tara C. Brady and George Panagiotopoulos (2017). An Integrated Nine-Step Approach to Managing Clinical Technology Risks. Biomedical Instrumentation & Technology: Sept./Oct. 2017, Vol. 51, No. 5, pp. 398-407.
